Brain extracellular space of the naked mole-rat expands and maintains normal diffusion under ischemic conditions
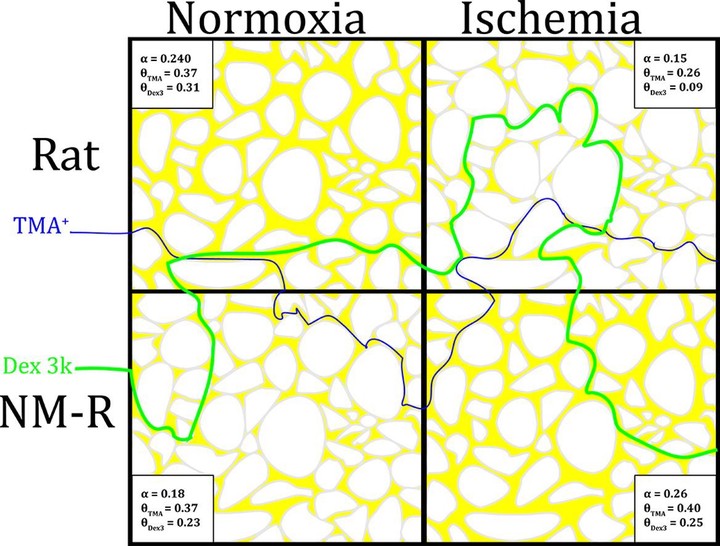 Comparison of naked mole-rat and rat brain extracellular space under normoxic and hypoxic consitions, as measured by iontophoretic optical imaging and real-time iontophoresis approaches.
Comparison of naked mole-rat and rat brain extracellular space under normoxic and hypoxic consitions, as measured by iontophoretic optical imaging and real-time iontophoresis approaches.
Abstract
Brain extracellular space (ECS) forms a conduit for diffusion, an essential mode of molecular transport between brain vasculature, neurons and glia. ECS volume is reduced under conditions of hypoxia and ischemia, contributing to impaired extracellular diffusion and consequent neuronal dysfunction and death. We investigated the ECS volume fraction and diffusion permeability of the African naked mole-rat (NM-R; Heterocephalus Glaber), a rodent with a remarkably high tolerance for hypoxia and ischemia. Real-Time Iontophoretic and Integrative Optical Imaging methods were used to evaluate diffusion transport in cortical slices under normoxic and ischemic conditions, and results were compared to values previously collected in rats. NM-R brains under normoxic conditions had a smaller ECS volume fraction than rats, and a correspondingly decreased diffusion permeability for macromolecules. Surprisingly, and in sharp contrast to rats, the NM-R ECS responded to ischemic conditions at the center of thick brain slices by expanding, rather than shrinking, and preserving diffusion permeabilities for small and large molecules. The NM-R thick slices also showed a blunted accumulation of ECS potassium compared to rats. The remarkable dynamic response of the NM-R ECS to ischemia likely demonstrates an adaptation for NM-R to maintain brain function in their extreme nest environment.